Produtos de anestesia Porto de Infusão Implantado
Porto de Infusão Implantado
O Infusão Port é um sistema de infusão intravenosa fechado que está totalmente implantado no corpo. Pode ser utilizado para infusão intravenosa a longo prazo, injeção intravenosa de solução nutricional e produtos sanguíneos, infusão de vários medicamentos e recolha de amostras de sangue em clínica.
CARACTERÍSTICAS
Especificidade do Modelo: As características podem variar entre modelos portuários específicos dentro da linha de produtos Baihe. Consulte sempre a rotulagem oficial do produto, as Instruções de Utilização (IFU) ou consulte diretamente a Baihe Medical para as especificações exatas de um modelo em particular.
Regulamentos: As funcionalidades e a disponibilidade podem diferir consoante as aprovações regulatórias regionais (por exemplo, NMPA na China, FDA nos EUA, CE na Europa).
Utilização profissional: A implantação e gestão requerem profissionais de saúde treinados.
- Implantado e Oculto: Sob a pele, sem partes externas.
Components:
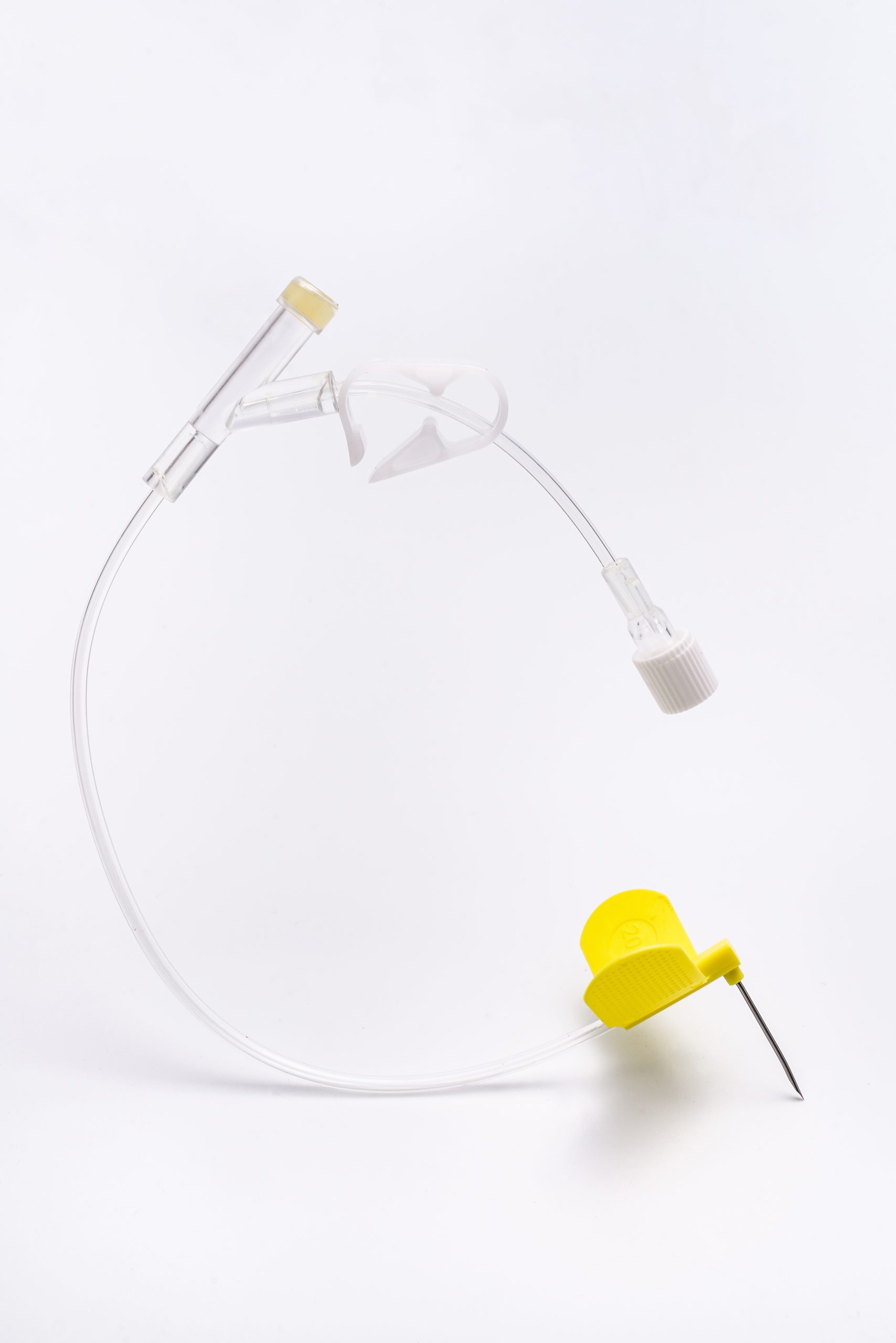
Reservatório do Porto (Corpo/Câmara): Uma pequena câmara em forma de disco feita de Titânio: Forte, biocompatível e segura para ressonância magnética. Contém um septo de silicone auto-selante no topo.
Cateter: Um tubo fino e flexível feito de material TPU único ligado ao reservatório da portaSepto Durável: Auto-selante, resiste a centenas de perfurações.
Acesso Central: Ponta do cateter numa veia central grande.
A longo prazo: Concebido para anos de uso.
Perfil Baixo: Visibilidade/volume mínimo.
Redução do risco de infeção: Comparado com cateteres externos.
Conveniência: Para terapia intravenosa frequente/prolongada e colheitas de sangue.
Acesso à Agulha: Requer uma agulha específica sem núcleo (Huber).

Materiais de Alta Qualidade
Corpo/Base do Porto: Reservatório do Porto (Corpo/Câmara): Uma pequena câmara em forma de disco feita de Titânio: Forte, biocompatível e segura para ressonância magnética. Contém um septo de silicone durável e auto-selante no topo. O titânio oferece excelente biocompatibilidade, compatibilidade com ressonância magnética (em condições específicas) e durabilidade. Septo: Feito de borracha de silicone de grau médico. Concebido para alta durabilidade e auto-selante após centenas a milhares de perfurações com agulha. Cateter: Feito principalmente de TPU, proporciona maior resistência à tração e à pressão.
Compatibilidade com Injetores de Energia (Compatibilidade CT)
Projetado e testado para suportar as altas pressões geradas durante a injeção de potência para tomografias computorizadas com contraste. Uma característica crítica para doentes oncológicos que frequentemente necessitam de imagiologia diagnóstica. Classificações de pressão específicas (300 psi, 5 mL/s).

Características de Segurança e Anti-Refluxo
Incorpore tecnologia de válvula anti-refluxo na ponta do cateter. Esta válvula: Abre-se sob pressão positiva durante a infusão. Fecha quando a infusão termina ou sob pressão negativa (como durante a aspiração sanguínea), prevenindo o refluxo sanguíneo para a ponta do cateter. Visa reduzir o risco de trombose e oclusão relacionadas com cateter, podendo diminuir as necessidades de manutenção (por exemplo, descargas de heparina menos frequentes).
Compatibilidade de Ressonância Magnética
A radiopacidade facilita a confirmação do posicionamento do cateter. As pontas das versões de múltiplos lúmenes são mais radiopacas, facilitando a confirmação do posicionamento da ponta fluoroscópica.
Disposable Endotracheal Tube
1. A principal matéria-prima do produto é o policloreto de vinilo (PVC) de grau médico;
2. O corpo do tubo é feito de material sensível ao calor, que se torna mole nas vias aéreas e não é fácil de danificar as vias aéreas. Tem dureza e elasticidade moderadas e não é fácil de dobrar;
3. O ângulo de bisel da ponta é o ângulo padrão, com transição suave da cabeça de fusão e sem rebarba;
4. A periferia do orifício de Murphy é lisa sem ângulo agudo, o que não é fácil de danificar a mucosa;
5. A ligação entre o punho e o corpo do tubo é suave, sem ângulo acentuado. O manguito é macio, comprido e largo, e a pressão por unidade de área é pequena para proteger a mucosa traqueal.
ESPECIFICAÇÕES
8Fr, 9Fr como tamanho padrão
Unidades individuais: Kits embalados em blister com papel Tyvek
Embalagem secundária: Papel ondulado de camada única.
Embalagem terciária: Cartão ondulado de parede dupla.
DOCUMENTS


