Productos de anestesia Puerto de infusión implantado
Puerto de infusión implantado
El Infusio Puerto es un sistema de infusión intravenosa cerrado que está completamente implantado en el cuerpo. Puede utilizarse para infusión intravenosa a largo plazo, inyección intravenosa de solución nutricional y productos sanguíneos, infusión de diversos fármacos y recogida de muestras de sangre en clínica.
CARACTERÍSTICAS
Especificidad del modelo: Las características pueden variar entre modelos específicos de puertos dentro de la línea de productos Baihe. Consulta siempre el etiquetado oficial del producto, las Instrucciones de Uso (IFU) o consulta directamente con Baihe Medical para conocer las especificaciones exactas de un modelo en particular.
Regulaciones: Las características y la disponibilidad pueden variar según las aprobaciones regulatorias regionales (por ejemplo, NMPA en China, FDA en EE. UU., CE en Europa).
Uso profesional: La implantación y el manejo requieren profesionales sanitarios formados.
- Implantado y oculto: Bajo la piel, sin partes externas.
Components:
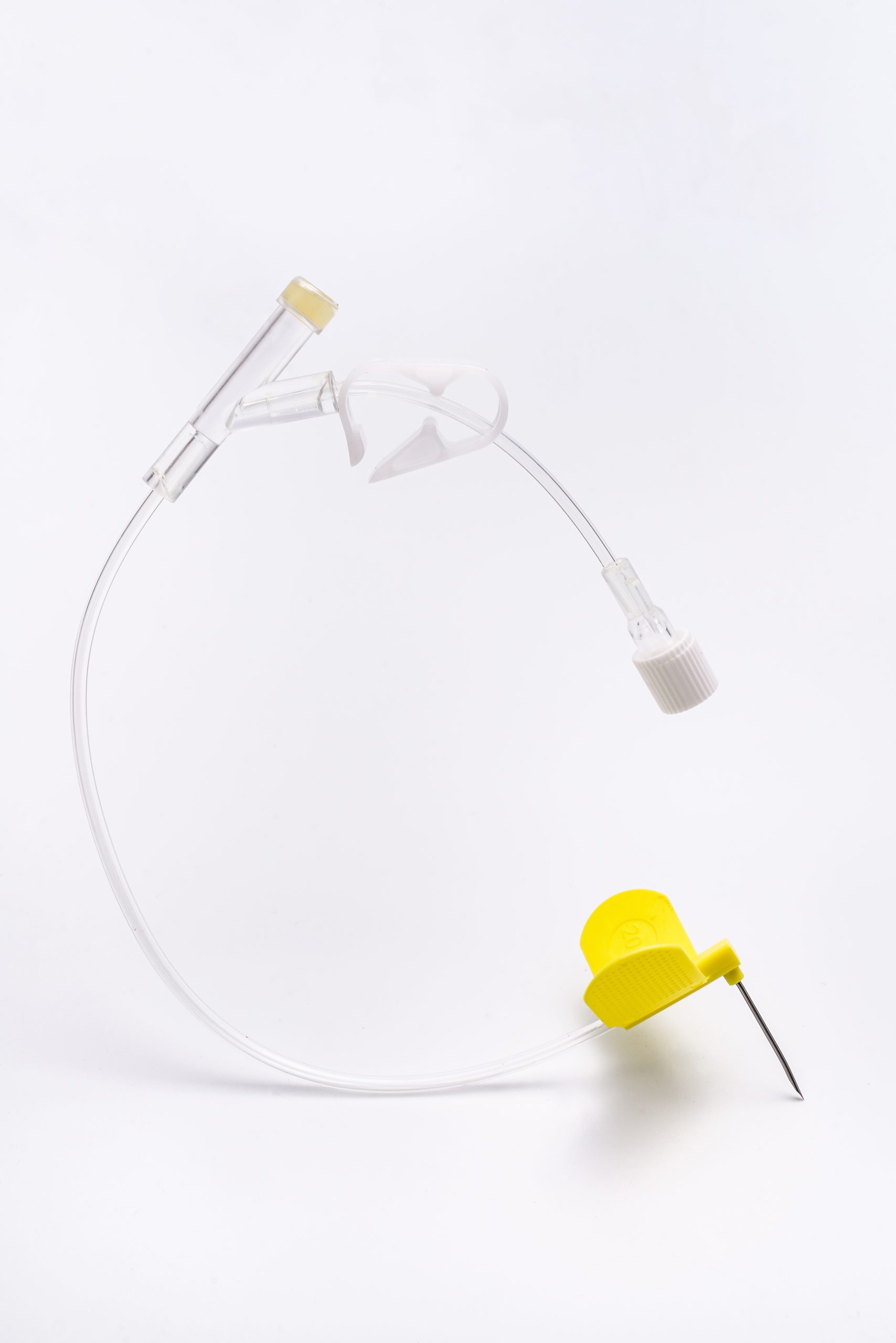
Depósito de Puerto (Cuerpo/Cámara): Una pequeña cámara en forma de disco hecha de titanio: Fuerte, biocompatible y segura para resonancias magnéticas. Contiene un tabique de silicona autosellante en la parte superior.
Catéter: Un tubo fino y flexible hecho de un material TPU único conectado al depósito del puertoTabique duradero: Autosellante, soporta cientos de pinchazos.
Acceso central: Punta del catéter en una vena central grande.
A largo plazo: Diseñado para años de uso.
Perfil bajo: visibilidad/volumen mínimo.
Menor riesgo de infección: En comparación con catéteres externos.
Comodidad: Para terapia intravenosa frecuente/prolongada y extracciones de sangre.
Acceso a la aguja: Requiere una aguja específica que no se descarone (Huber).

Materiales de alta calidad
Cuerpo/Base del puerto: Depósito del puerto (Cuerpo/Cámara): Una pequeña cámara en forma de disco hecha de titanio: Fuerte, biocompatible y segura para resonancias magnéticas. Contiene un tabique de silicona duradero y autosellante en la parte superior. El titanio ofrece una excelente biocompatibilidad, compatibilidad con resonancia magnética (bajo condiciones específicas) y durabilidad. Septum: Hecho de caucho de silicona de grado médico. Diseñado para una alta durabilidad y autosellante tras cientos o miles de pinchazos con aguja. Catéter: Principalmente fabricado en TPU, proporciona mayor resistencia a la tracción y a la presión.
Compatibilidad con inyectores de potencia (compatibilidad CT)
Diseñado y probado para soportar las altas presiones generadas durante la inyección de potencia para tomografías computarizadas con contraste mejorado. Una característica crítica para los pacientes oncológicos que con frecuencia requieren imágenes diagnósticas. Presión específica (300 psi, 5 mL/seg).

Características de seguridad y antireflujo
Incorpora tecnología de válvula antireflujo en la punta del catéter. Esta válvula: Se abre bajo presión positiva durante la infusión. Se cierra cuando la infusión se detiene o bajo presión negativa (como durante la aspiración sanguínea), evitando el reflujo sanguíneo hacia la punta del catéter. Tiene como objetivo reducir el riesgo de trombosis y oclusión relacionadas con catéteres, lo que podría disminuir los requisitos de mantenimiento (por ejemplo, lavados de heparina menos frecuentes).
Compatibilidad con resonancia magnética
La radiopatía facilita la confirmación de la colocación del catéter. Las puntas de la versión multilúmen son más radiopacas, lo que facilita confirmar la colocación de la punta fluoroscópica.
Disposable Endotracheal Tube
1. La materia prima principal del producto es cloruro de polivinilo (PVC) de grado médico;
2. El cuerpo del tubo está hecho de material sensible a la temperatura, que se ablandará en la vía aérea y no dañará fácilmente la vía aérea. Tiene una dureza y elasticidad moderadas y no es fácil de doblar;
3. El ángulo de bisel de la punta es el ángulo estándar, con transición suave de la cabeza de fusión y sin rebaba;
4. La periferia del orificio de Murphy es lisa sin ángulo agudo, lo que no es fácil de dañar la mucosa;
5. La conexión entre el manguito y el cuerpo del tubo es suave sin ángulo pronunciado. El manguito es blando, largo y ancho, y la presión por unidad de área es pequeña para proteger la mucosa traqueal.
ESPECIFICACIONES
8Fr, 9Fr como tamaño estándar
Unidades individuales: Kits empaquetados en blíster con papel Tyvek
Embalaje secundario: papel corrugado de una sola capa.
Embalagem terciária: Cartão ondulado de parede dupla.
DOCUMENTS


